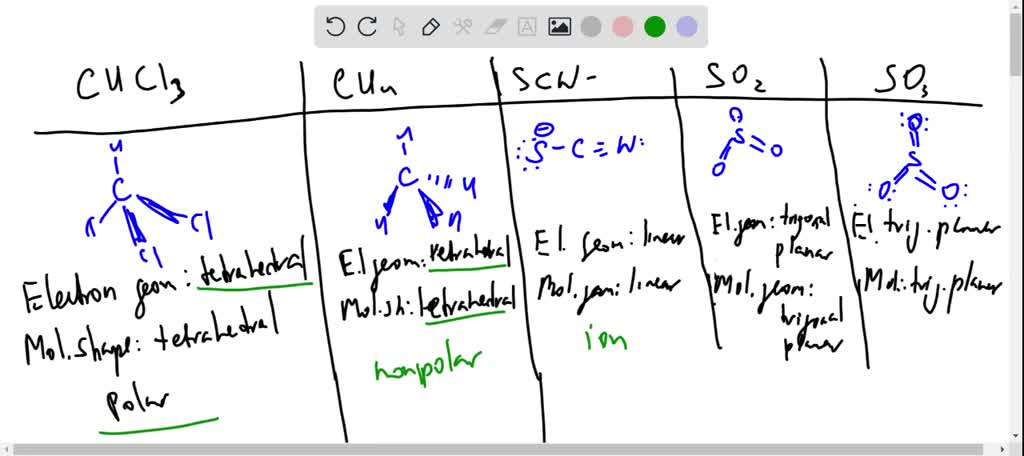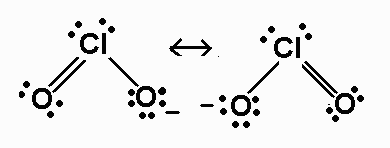
SOLVED: Classify the following bonds as ionic, polar covalent, or covalent and give your reasons a) the CC bond in H3CCH3 b) the KI bond in KI c) the ClO bond in
An Integrative Synthetic, Spectroscopic, and Computational Study of the Free Radical Chlorine Dioxide and its Interactions with