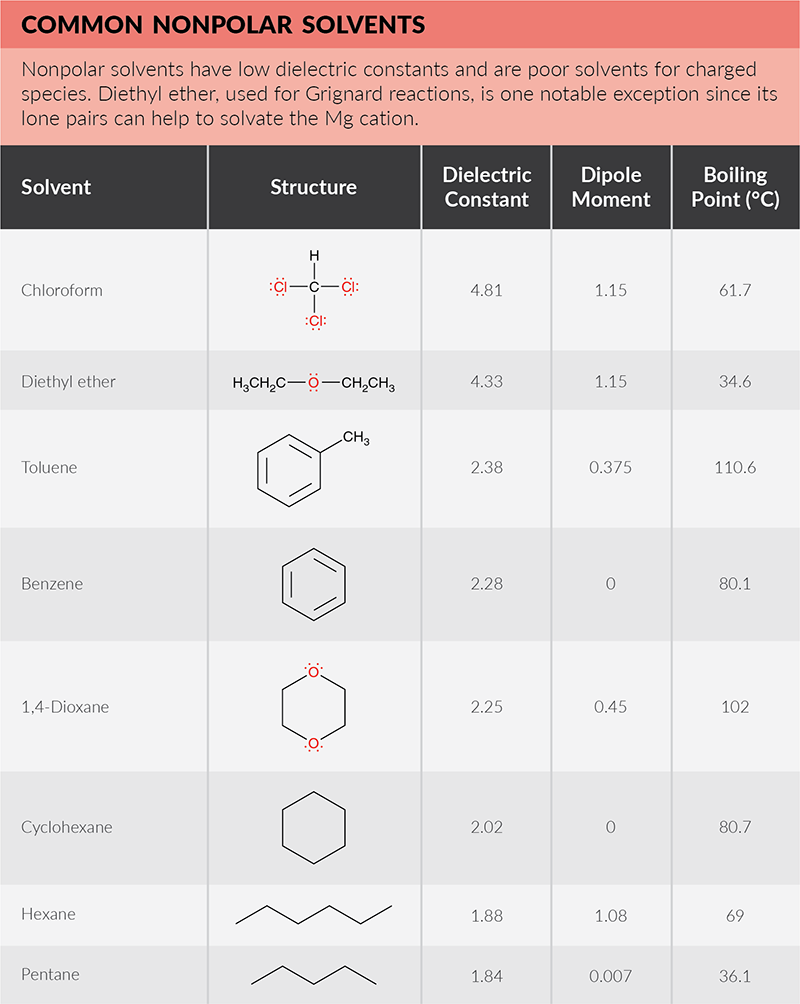
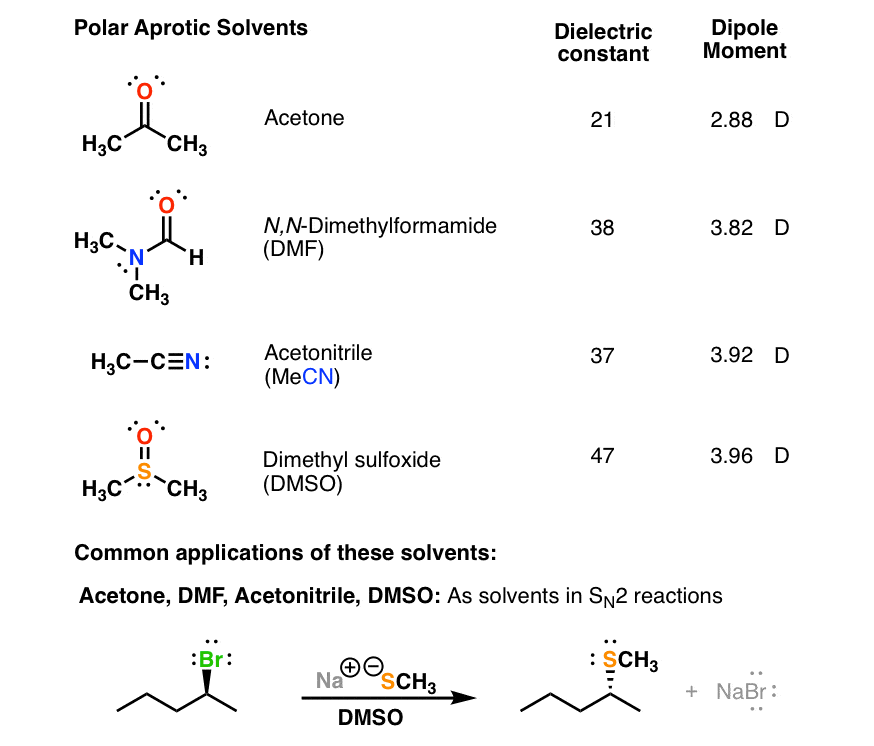
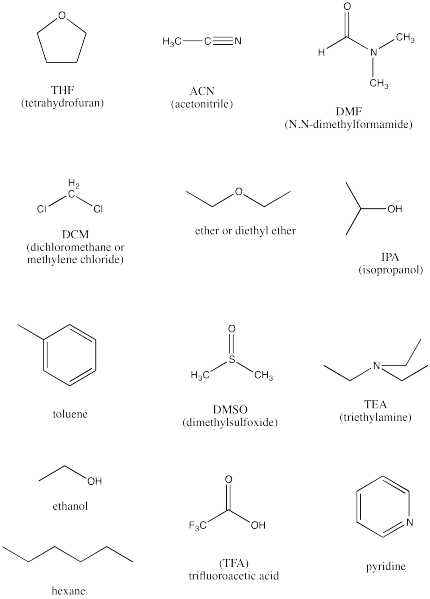
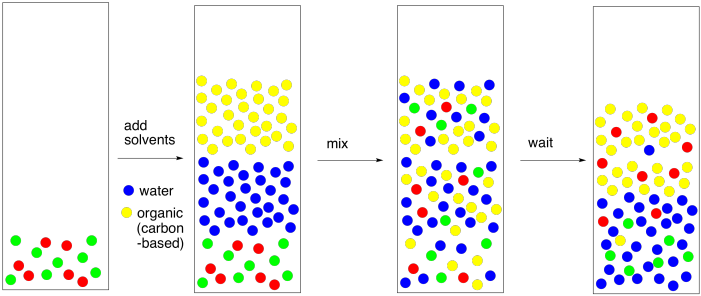
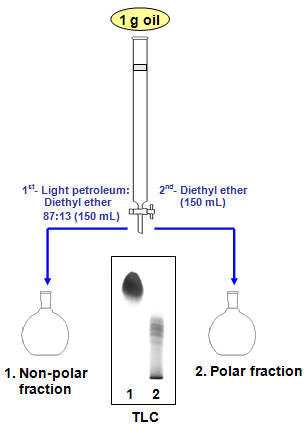
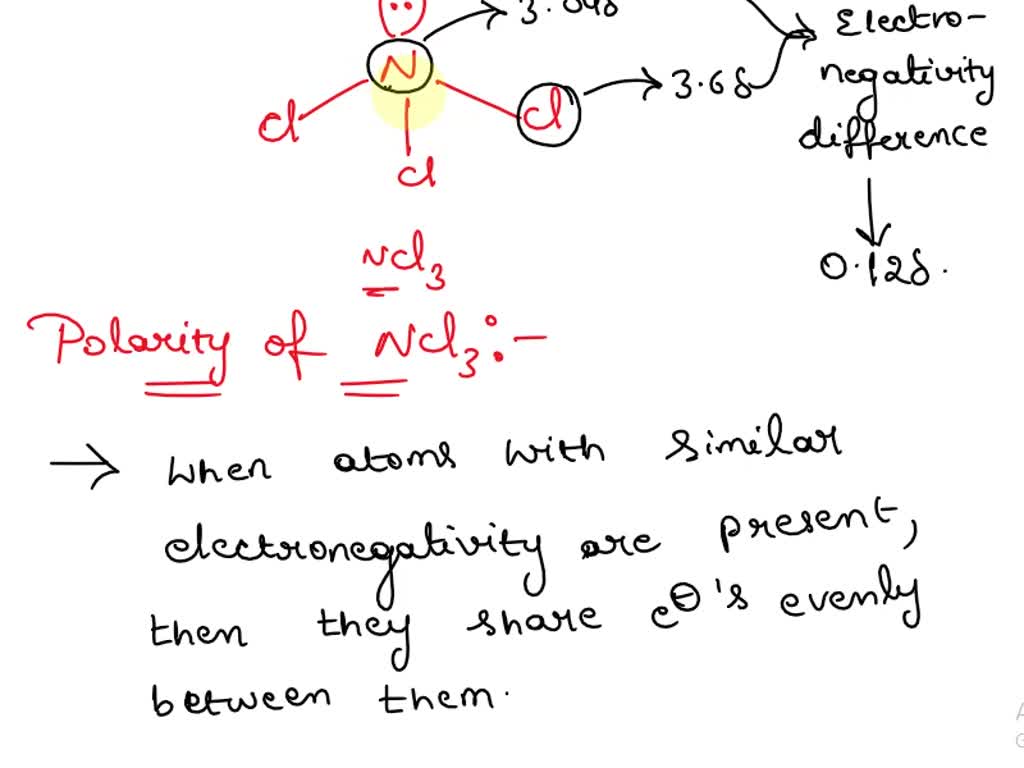
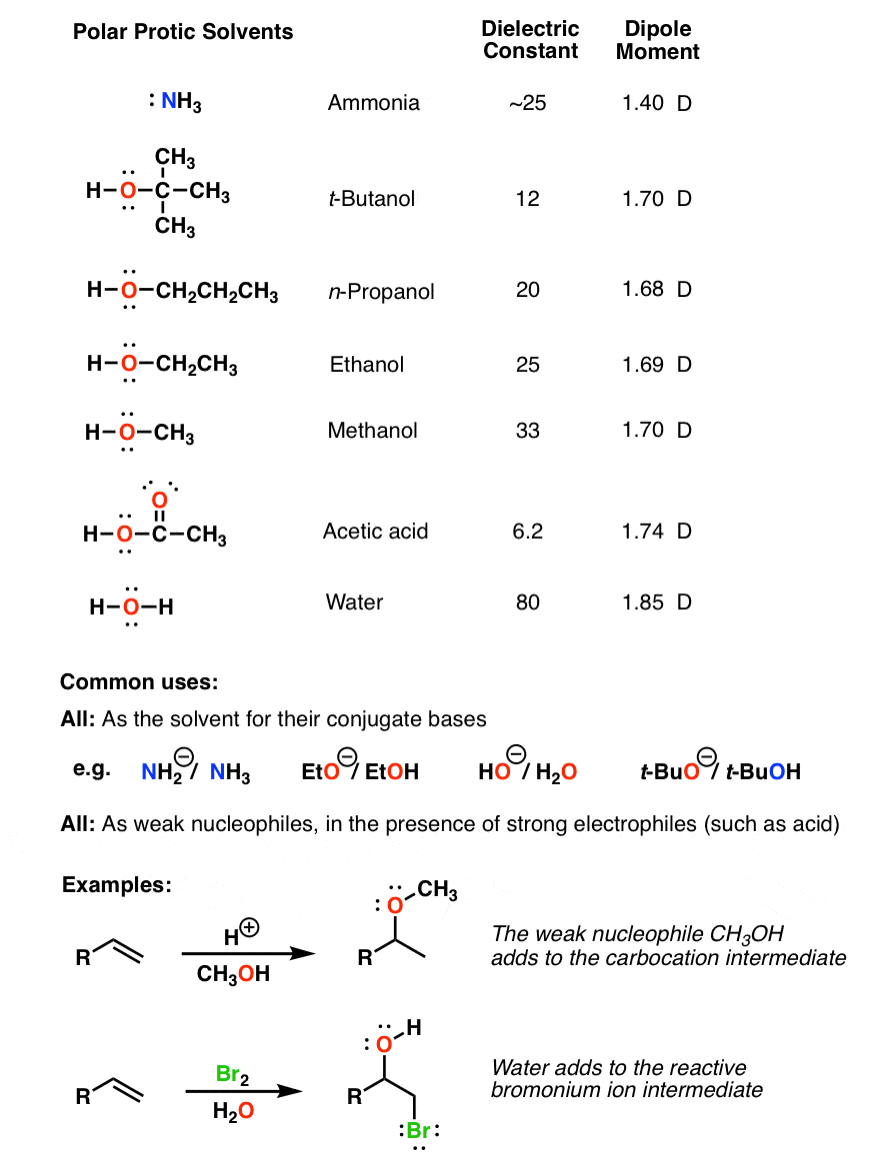
Arrange the following solvents in order of increasing polarity: a) ethanol b) ethyl acetate c) petroleum ether d) toluene e) acetone | Homework.Study.com

How can benzoic acid dissolve into diethyl ether since benzoic acid is polar and diethyl ether is non-polar? | Homework.Study.com