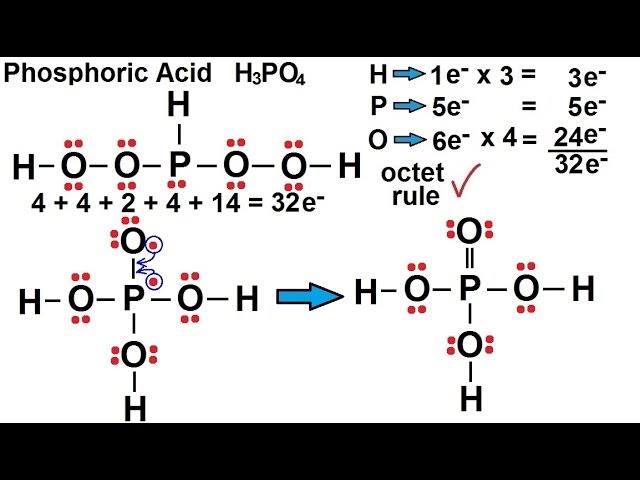
Phosphorous acid, H_3PO_3, has the structure (HO)_2PHO, in which one H atom is bonded to the P atom, and two H atoms are bonded to O atoms. For each bond to an

SOLVED: 1.what is lattice energy? 2.what is solvation energy?how it effects the solubility of electrovalent compound into polar and non polar solvent? 3.give the electron dot formula anddash formula of following= BeCl2
Phosphoric acid, dimethyl 4-nitrophenyl ester (CAS 950-35-6) - Chemical & Physical Properties by Cheméo